1 - B3 Radiochromic Film Dosimeter
B3 radiochromic film dosimeters are used for absorbed dose measurements in radiation processing. When irradiated, a chemical reaction occurs within the film causing a change in the visible light spectrum; this change is directly related to the quantity of absorbed dose the film receives and is consistent and quantifiable. See Figure 1.
Due to the film’s small thickness and low mass, it is an excellent dosimeter for measuring dose to products irradiated by electron beam, X-ray, and gamma technologies. Examples of end uses of such technologies include medical device sterilization, curing of inks and coatings and the decontamination of food or food packaging. Dosimeters are commonly used for mapping of dose distribution during qualification of these irradiation processes and routinely monitoring the process output, dose magnitude, for conformity assessment and process control purposes.
Intended Use
B3 film dosimeters are a part of a radiochromic film dosimetry system, as defined in ASTM 52628. B3 dosimeter products are intended to be used a part of a dosimeter measurement system that includes:
-
B3 film dosimeters
-
Reader (spectrophotometer)
-
Heat treatment device (incubator)
-
Dosimeter measurement process and procedures.
GEX B3 dosimeter products
GEX manufactures a variety of B3 dosimeter products, ideal for routine dosimeter measurements, research and testing of Electron Beam, X-Ray and Gamma irradiation processes. GEX B3 dosimeter products utilize the B3 radiochromic film and are designed to meet current industry guidelines.
2 - Calculating Dose
The response (R) of the dosimeter to ionizing radiation is defined in terms of optical absorbance (Ai) per unit thickness (t), also referred to as the “specific absorbance”,
R = Ai/t
Users may occasionally subtract the unirradiated, original absorbance (Ao), sometimes called “background Absorbance”, from the Ai to derive the “net specific absorbance”, such that R = (Ai-Ao)/t
For B3 dosimeters, the Ao is typically ignored for the following reasons:
-
Ao is stable (unchanging) over a long period of time.
-
B3 dosimeter thickness is both thin and uniform and the measurement of the Ao cannot help the user to resolve the small variances in thickness between dosimeters in the same batch and average thickness group.
-
Since there is no technical gain in subtracting Ao, GEX recommends that the user ignore the Ao in any calculation of R, and to use the simple response calculation of R = Ai/t
The relationship of R with absorbed dose (D) is expressed as Ai/t = R = ƒ(D) where the dosimeter response is a function of the quantity of absorbed dose received by the dosimeter. To calculate absorbed dose from the dosimeter’s measured Absorbance,
Ai/t = R = f(D)
The GEX DoseControl Dosimetry System automatically calculates absorbed dose (D) for each dosimeter measurement.
3 - Response
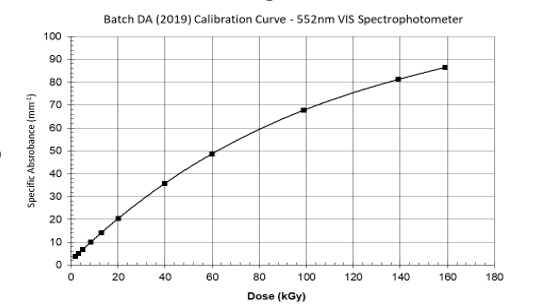
The response (R) has a slope of 2.0 at approximately 2.0 kGy, reducing to a slope of 1.0 near 25 kGy, and a slope near 0.6 at approximately 100 kGy.
Response Measurement Reproducibility – The user should be able to achieve the following percentage coefficient of variation (C.V.%) for repeated measurement of the same dosimeter:
1.5% or better at 1.0 kGy
1.0% or better at 10.0 kGy
0.5% or better at 20.0 kGy and higher
4 - Resolution
B3 has a calculated dose resolution of approximately 0.1 kGy at 25 kGy. Every 0.001 Ai change results in a change of approximately 0.1 kGy.
5 - Practical Dose Range
The dose range of the B3 film is approximately <1 kGy to 160 kGy. Some users have been successful in using the product above and below these values. Saturation of response begins near 150 kGy with full response saturation occurring at a dose of approximately 165 kGy or a specific absorbance of approximately 90.0 A*mm-1 (approximately 1.700 A) under normal temperature conditions.
3The low dose range may be extended by special techniques while the high dose range is situationally dependent on the irradiation conditions. The user should consider measurement uncertainty when deciding on the practical range. Some users have been successful in using the product above and below these values.
6 - Dosimeter Batch Calibration (also called ‘dosimetry system calibration’)
The dosimetry system must be calibrated to achieve dose values traceable to an international standard (per ASTM 52628 Standard Practice for Dosimetry in Radiation Processing).
NOTE: Most radiation process applications require dose measurements to be traceable to a recognized national standard. There are a few applications where only relative dose measurements are carried out, for example, beam width measurements, that may not require traceability (per ASTM 51261).
More information:
-
Complete information, guidance, technical reports: Dosimeter Batch Calibration (Dosimetry System Calibration).
-
Specific B3 dosimeter batch calibration overview: B3 Dosimeter Batch Calibration
7 - Dosimeter Measurement Uncertainty
All measurements of absorbed dose (D) must be accompanied by an estimate of uncertainty. All components of uncertainty should be included in the estimated uncertainty, including those arising from calibration (dosimeter batch calibration), dosimeter reproducibility, instrument stability and the effect of influence quantities. See complete information here: Dosimetry System Calibration - Developing and Using Uncertainty Statements.
Disclaimer -The information contained in this document is provided “as is” and is not a substitute for the user’s professional judgement. It is provided as a convenience to those using products provided by GEX Corporation who have sufficient technical skills to evaluate and properly apply the information in this document. It is the responsibility of the user of this document to ensure that the information in this document, and the use of such information, is accurate, complete, applicable to the product, suitable for the user’s purposes, and in compliance with all laws and regulations. GEX Corporation believes the information provided in this document is accurate and reliable as of the time of writing, but it undertakes no obligation to update or correct this document. GEX Corporation may, but is not required to, make changes to this document at any time without notice. By using the information in this document, the user represents and warrants that he or she has the skills necessary to properly understand and apply this information and that he or she will comply with all applicable laws and regulations including, without limitation, those relating to medical devices, pharmaceutical products, or other applicable industries. The user assumes all risks associated with using this information and any results or output resulting from the application of this information to GEX Corporation’s products. The user agrees not to hold GEX Corporation liable for any errors or omissions contained within.
